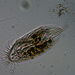
Photos / Sounds
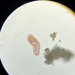
What
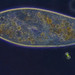
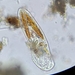
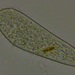
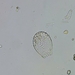
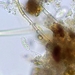
Genus GaleriporaObserver
someplantDescription
Magnification of photos: 100×, 200×, 200×, 400×, 400×, 400×, 400×, 400×, 600×, 600×
Habitat: organic debris and water squeezed from Ceratophyllum, growing in a slow-moving part of a brook.
Photo taken with a Celestron PentaView Digital Microscope. According to their website, the FOV (i.e. the diagonal width) at 100× is 600 µm.
Photos / Sounds
What
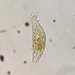
Hypotrichs (Subclass Hypotrichia)Observer
jordi_mvDescription
A ciliate with a red pigmentation
400X
Eyepiece: WF10X DIN/18MM
Objective: S40/0.65 - 160/0.17
Sample of river water collected at the shore
Photos / Sounds
What
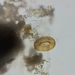
Stentor roeseliiObserver
jordi_mvDescription
400X, (The last photo was taken at 100X)
Eyepiece: WF10X DIN/18MM
Objective: S40/0.65 - 160/0.17
Sample of river water collected at the shore
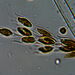
Photos / Sounds
What
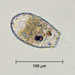
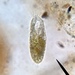
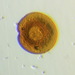
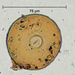
Histriculus histrioObserver
someplantDescription
Magnification of photos: 200×, 200×, 200×, 200×, 200×, 200×, 200×, 400×, 400×, 400×, 400×, 400×
Habitat: water and organic debris from a swamp, collected with Chaetophoropsis. Associated plants: Osmunda spectabilis, Clethra alnifolia
Photo taken with a Celestron PentaView Digital Microscope. According to their website, the FOV (i.e. the diagonal width) at 200× is 300 µm.
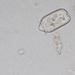
Photos / Sounds
What
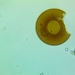
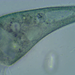
Genus DinobryonObserver
someplantDescription
Magnification of photos: 400×, 600×, 600×, 600×
Habitat: water and organic debris from a swamp, collected with Chaetophoropsis. Associated plants: Osmunda spectabilis, Clethra alnifolia
Photo taken with a Celestron PentaView Digital Microscope. According to their website, the FOV (i.e. the diagonal width) at 400× is 150 µm.
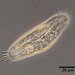
Photos / Sounds
What
Stentor igneusObserver
someplantDescription
Magnification of photos: 200×, 200×, 200×, 200×, 400×, 400×, 400×, 400×, 400×, 600×, 600×, 600×, 600×
Habitat: water and organic debris from a swamp, collected with Chaetophoropsis. Associated plants: Osmunda spectabilis, Clethra alnifolia
Photo taken with a Celestron PentaView Digital Microscope. According to their website, the FOV (i.e. the diagonal width) at 200× is 300 µm.
Photos / Sounds
What
Genus ThuricolaObserver
lassa8Description
Two trumpet shaped cells with a circle (?) of cilia at the top. No stalk visible like with Vorticella-like cells. Contracted into two small balls now and then before opening up again.
Attached to algae collected in Chesapeake Bay.
Photos / Sounds
What
Order SessilidaObserver
lassa8Description
Collected with algae from Chesapeake Bay.
It mostly spun in slow circles in all directions. It may have had tufts of cilia on its flat end to go with the ring of cilia on its pointed end.
Third file is a small GIF taken at 120 fps, so it's slower than life speed.
Forgot to adjust my condenser after moving from 5x to 40x objective, so resolution is low.
Photos / Sounds
What
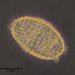
Anigsteinia salinaraObserver
peptolabDescription
Anigsteinia salinara (Florentin) Isquith,1968 from the intertidal benthos of marine estuary Acabonac Harbor at Landing Lane launching ramp. The sample was fed with boiled wheat seed. Imaged in Nomarski DIC on Olympus BH2S using SPlanapo 40 0.90 and SPlan 100 1.25 oil with oiled condenser plus variable phone camera cropping on Samsung Galaxy S9+. The cells range from 160 up to 210 um in length. The distinctive macronuclear apparatus consisting of numerous irregularly clustered spherical nodules distinguish this species from A. clarissima, a species I have found often in samples of this and other estuaries. In fact, A. salinara is the only member of the genus to have this type of macronuclear apparatus; most are moniliform and one, A. candida, has 2-3 times as many nodules as A. salinara arranged in irregular linear arrays down the center of the cell. Also, A. salinara has a mid-region that is often swollen compared to the anterior end.
"Anigsteinia salinara (Florentin) Isquith,1968 Synonymy: Spirostomum salinarum Florentin, 1899 . Blepharisma salinarum Kahl, 1928 Non: B. salinarum described by Dragesco in 1963. This form does not seem to be a member of Anigsteinia. Florentin (1899) described this form from extremely saline waters (65 g/l). It ranges in size from 160-360 um. The organism is colorless to faintly yellow, non-contractile, and rigid. The somatic cilia are short and numerous. The peristome extends about one-half the body length, starting at an anterior beak. There is a well-developed AZM; the undulating membrane extends about one-fifth the peristomal length. The cytopharynx is quite short. The macronucleus consists of 50-75 spherical fragments of 2.5-5.0 um diameter each; Florentin (1899) was also able to distinguish micronuclei. The collecting canal feeding the contractile is quite obvious. The mid-region of the organism tends to appear quite swollen. In 1928, Kahl transferred this organism from Spirostomum to Blepharisma, because it had an undulating membrane and was non-contractile. He felt that Florentin had placed it in the former species, since that author had not observed the undulating membrane. Kahl (1928) further questioned Florentin's placement of the organisms, since it was non-contractile. The form occurs in strongly saline waters and also weakly brackish ones (Kahl 1928)" (1).
"Anigsteinia is characterized by being a quite elongate spirostomatid, with a lacunar contractile vacuole system. On the left margin of the peristome there is a well-developed adoral zone of membranelles (AZM), and on the right margin a relatively inconspicuous undulating membrane is present. Slight body contraction (without body torsion) is apparent in some species. The somatic ciliature of Anigsteinia is basically the same as that found in other spirostomatids. The ciliary rows run the length of the body. The rows on the left side of the body, in the region of the oral groove, turn towards and terminate at the AZM. The somatic rows on the right side of the organism run straight to the anterior tip of the organisms. The genus was originally established by Isquith in 1968" (1).
"In essence, this genus is distinguished from Blepharisma by the possession of the collecting canal for the contractile vacuole, a more elongate body, a less conspicuous undulating membrane, and somatic contractility in some species. It is distinguished from Spirostomum by the lack of body torsion during contraction, and the presence of an undulating membrane. A comparison of the three genera is provided in Table 1. Phylogenetically, Anigsteinia appears to fit between the other two genera. The closeness of these three is well illustrated by Anigsteinia salinara. It was originally named Spirostomum salinarum by Flo r entin in 1899, but it was transferred to Blepharisma by Kahl in 1928. Since the two genera are quite distinct, if A. salinara were not an intermediate between the two, such confusion would not have been possible. The creation of the intermediate genus, Anigsteinia, should eliminate any further taxonomical confusion as to where Florentin's organism belongs as well as several closely related species" (1).
"Blepharisma (Spirostomum) salinarum FLORENTIN, 1899 Size 150-250 um, according to FLORENTIN up to 360 um. The French researchers had the three-sided undulating membrane, which usually protrudes into the field of vision. It is only about 1/5 as long as the peristome which is about 1/2 body length. Shape maybe after the middle it usually expands somewhat, flattened (2: 1). Ecoplasm coarse, shiny, extensively furrowed, with one each fibril in the intermediate stripe, colorless or yellowish. Cilia dense and short. Contractile vacuole terminal. Nucleus in 70-100 very small disordered chunks the middle of the body with many very small Mi. Slowly sliding or rotating; especially eats rhodobacteria. Euryhaline, polysaprobic, inland salt sites Lorraine, Oldesloe, brackish water near Cuxhaven, Kiel. Common to numerous" (2).
- Isquith IR, Repak AJ. 1974. The systematics of the genus Anigsteinia Isquith, 1968. Acta Protozoologica 15: 193 - 200.
- Kahl A. 1932. Urtiere oder Protozoa I: wimpertiere oder Ciliata (Infusoria) 3. Spirotricha. DieTierwelt Deutschlands Pt. 25 (year 1932): 399 - 650.
Photos / Sounds
What
Arcella costataObserver
douchDescription
In estuary. Bright-field microscopy.
Photos / Sounds
What
Euglypha ciliataObserver
ilsignorvegaDescription
inside wet moss at the edge of a small lake
Photos / Sounds
What
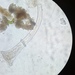
Stentor coeruleusObserver
peptolabDescription
Stentor coeruleus Ehrenberg, 1830, showing a rare and unusual bicoloration which is likely environmentally produced from a sample of the riverside benthos of the freshwater portion of the estuarine river Peconic River. The sample was fed with boiled wheat seed. Imaged in Nomarski DIC (and brightfield at 40x and 100 x) on Olympus BH2S using Splan 4, Splan 10, SPlanapo 20 0.70, Splanapo 40 0.95, and SPlan 100 1.25 oil objectives plus variable phone camera cropping on Samsung Galaxy S9+.
The cells so far showed only swimming forms and measure from 520 up to 800 um in length; I have not seen them anchor and fully extend in the manner of S. coeruleus. This size range is rather small for Stentor coeruleus. The spiral of the oral pit appears dark at low magnification and red with higher magnification and stands out starkly from the turquois blue body. At high magnification, one can see mainly turquoise-colored pigmented pellicular granules with fewer smaller red granules between the somatic kineties but red granules are strikingly concentrated in the buccal spiral pit.
I first considered S. introversus Tartar 1958 based on the mentioned close resemblence to S. coeruleus in most characters- turquois blue color without symbiont algae, moniliform macronculeus, smaller size (1,2). My misdiagnosis was largely based on a paper by Kim et al 2020 (3) who redescribed a brackish population which they called S. introversus based largely on the presence of two colors of granules, turquoise blue (somatic) and red (3). However, this feature is not mentioned anywhere with regard to S. introversus. In fact Foissner et al describe S. introversus TARTAR 1958: "which is described in the USA and is identical in granulation and nuclear apparatus, is slightly smaller (up to 450 um), has a brown-yellow cytoplasm and everts its front end characteristically during contraction" (1). Perhaps more importantly, Kim et al 2020 (3) did not illustrate the main defining character of S. introversus in their population as defined by Tartar 1958 (4): the eversion of the buccal field, and that "border of recurved ectoplasm" which he calls "somewhat scalloped". Indeed, the buccal field of my population is that of garden variety S. coeruleus.
So, how to resolve these differential diagnostic discrepancies. Foissner and Wölfl 1994 included S. introversus in their review of the genus: "Stentor introversus Tartar, 1958. This species matches most characters of S.coeruleus. It is, however, smaller [450 um, up to 580 um, according to Jones (1974); weak character] and the entire zone of adoral membranelles cannot only contract, but also retract. We recognize this form as a distinct species because heteroplastic graft combinations between S.introversus and S.coeruleus did not survive (Tartar, 1958), and because Tartar was an expert on S.coeruleus making it unlikely that he was mistaken" (2). Kim et al found Stentor inversus in Korea, ostensibly the first report of the species since Tartar described it in 1958. "Stentor introversus Tartar, 1958 Diagnosis. Extended body size in vivo 500-600×200 250 μm and trumpet-shaped, contracted body size 250 305×200-250 μm in vivo (n=10) and pouch-shaped with retracted buccal field; cytoplasm colorless; 2 kinds of cortical granules densely arranged in between somatic kineties, larger granules turquoise and 0.4-0.5 μm in diameter, smaller granules reddish and 0.2-0.3 μm in diameter; 250 adoral membranelles; 60-73 somatic kineties; 16-20 buccal kineties including 1 peristomial kinety; moniliform macronucleus with 9-14 nodules. Distribution. U.S.A. and Korea. Remarks. Stentor introversus was not reported since the original description by Tartar (1958) from USA. It differs from the most similar species, S. coeruleus (Pallas, 1766) Ehrenberg, 1831, in the extended body size (500-600 μm vs. 1-2 mm), buccal field retracted when contracted (present vs. absent), and the number of types of cortical granules (2 vs. 1) (Foissner et al., 1992; Foissner and Wölfl, 1994) (3).
In evaluating my diagnosis of this population as S. introversus, Bruce Taylor made me aware of some interesting research on S. coeruleus by Tartar 1972 (5), who Foissner acknowledges as an " expert on Stentor coeruleus" (2). In Tartar's experiments, "after a few days in caffeine solution the dark blue-green ciliate Stentor coeruleus loses all visible pigmentation. The cells remain viable and transparent if the solution is then diluted. In time, however, a light pink color is regenerated in maintenance concentration of the drug. In caffeine-free medium the major dark green pigment is regenerated" (5). Tartar feels that the pink coloration was present all the while: "at first colorless, the animals acquired a light pink color, sometimes two or three days after bleaching or sometimes only three or four weeks later, depending on the strain. That this pigment was regenerated and not a new pigment developed under the influence of caffeine is indicated by the fact that at weaker bleaching concentrations of the drug the slower decoloration removed the green but left a pink color from the start, as if a native pink pigment had been unmasked by loss of the green one" (5).
The presence of two native pigments, pink-red and blue, in S. coeruleus is also supported by the work of Moller 1962 (6). Tartar 1972 (5) pointed out: "These two pigments, now demonstrable by separate regeneration of the pink, probably are those discovered by Moller 1962 using extraction procedures on S. coeruleus: a pink one he called stentorin-1 which was alcohol soluble, and a predominating blue-green pigment - stentorin-2, not extractable with alcohol" (5).
So my population is almost certainly an environmentally induced phenotype of Stentor coeruleus which, due to some unknown environmental stimulus, has developed a strikingly visible pink-red coloration of the oral spiral while retaining its usual predominant turquoise granulation elsewhere. The presence of two different colored granules in S. introversus sensu Kim et al 2020 is also explainable in this way. While my population of S. coeruleus is smaller, so far observed at 540-800 um, I have not yet observed sessile feeding forms which may well reach the 1-2 mm length reported for S. coeruleus.
- Foissner,W., Berger,H. and Kohmann,F. (1992) Taxonomische und ökologische Revision der Ciliaten des Saprobiensystems-Band II: Peritrichia, Heterotrichida, Odontostomatida. Informationsberichte des Bayer. Landesamtes für Wasserwirtschaft, May 1992, pp. 351-4
- Revision of the genus Stentor Oken (Protozoa, Ciliophora) and description of S.araucanus nov. spec. from South American lakes. W.Foissner and S.Wölfl. Journal of Plankton Research Vol.l6 no.3 pp.255-289. 1994
- Taxonomy of 16 indigenous ciliate species (Protozoa, Ciliophora) from South Korea. Ji Hye Kim, Atef Omar, Ji Hye Moon and Jae-Ho Jung. Journal of Species Research 9(4):427-442, 2020
- Tartar, V. (1958) Stentor introversus, n. sp. J. Protozool., 5, 93-95.
- Caffeine bleaching of Stentor coeruleus. Vance Tartar. Journal of Experimental Zoology Volume181, Issue 2 August 1972 pp 245-25.
- Moller, K. M. 1962 On the nature of stentorin. C. R. Lab. Carsberg, 32: 471-498.
Photos / Sounds
What
Stentor multiformisObserver
manuel_helsinkiDescription
Baltic sea sample with algae and sand collected from a beach and observed within few days of collection.
Stats
- 7608